IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
Perforations and Fistulas: Gastrointestinal (GI) perforations and fistulas, including fatal cases, were reported in 3% and 1% of COMETRIQ-treated patients (N=214), respectively. Non-GI fistulas, including tracheal/esophageal fistulas, including fatal cases, were reported in 4% of COMETRIQ-treated patients. Monitor patients for symptoms of perforations and fistulas, including abscess and sepsis. Discontinue COMETRIQ in patients who experience a Grade 4 fistula or a GI perforation.
Hemorrhage: Severe and fatal hemorrhage occurred with COMETRIQ. The incidence of Grade ≥3 hemorrhagic events was higher in COMETRIQ-treated patients compared with placebo (3% vs 1%). Discontinue COMETRIQ for Grade 3 or 4 hemorrhage. Do not administer COMETRIQ to patients with a recent history of hemorrhage, including hemoptysis, hematemesis, or melena.
Thrombotic Events: COMETRIQ treatment resulted in an increased incidence vs placebo of venous thromboembolism (6% vs 3%) and arterial thromboembolism (2% vs 0%). Discontinue COMETRIQ in patients who develop an acute myocardial infarction or arterial or venous thromboembolic events that require medical intervention.
Impaired Wound Healing: Wound complications have been reported with COMETRIQ. Withhold COMETRIQ for at least 3 weeks prior to elective surgery. Do not administer COMETRIQ for at least 2 weeks after major surgery and until adequate wound healing is observed. The safety of resumption of COMETRIQ after resolution of wound healing complications has not been established.
Hypertension and Hypertensive Crisis: COMETRIQ treatment resulted in an increased incidence of treatment-emergent hypertension vs placebo (61% vs 30%). Do not initiate COMETRIQ in patients with uncontrolled hypertension. Monitor blood pressure regularly during COMETRIQ treatment. Withhold COMETRIQ for hypertension that is not adequately controlled with medical management; when controlled, resume COMETRIQ at a reduced dose. Discontinue COMETRIQ for severe hypertension that cannot be controlled with anti-hypertensive therapy and for hypertensive crisis.
Osteonecrosis of the Jaw (ONJ): ONJ occurred in 1% of COMETRIQ-treated patients. ONJ can manifest as jaw pain, osteomyelitis, osteitis, bone erosion, tooth or periodontal infection, toothache, gingival ulceration or erosion, or persistent jaw pain or slow healing of the mouth or jaw after dental surgery. Perform an oral examination prior to initiation of COMETRIQ and periodically during COMETRIQ treatment. Advise patients regarding good oral hygiene practices. Withhold COMETRIQ treatment for at least 3 weeks prior to scheduled dental surgery or invasive dental procedures, if possible. Withhold COMETRIQ for development of ONJ until complete resolution.
Diarrhea: Diarrhea occurred in 63% of patients treated with COMETRIQ. Grade 3 to 4 diarrhea occurred in 16% of patients treated with COMETRIQ. Withhold COMETRIQ until improvement to Grade 1 and resume COMETRIQ at a reduced dose for intolerable Grade 2 diarrhea, Grade 3 diarrhea that cannot be managed with standard antidiarrheal treatments, or Grade 4 diarrhea.
Palmar-Plantar Erythrodysesthesia (PPE): PPE occurred in 50% of patients treated with COMETRIQ and was severe (Grade 3) in 13% of patients. Withhold COMETRIQ in patients who develop intolerable Grade 2 PPE or Grade 3 PPE until improvement to Grade 1; resume COMETRIQ at a reduced dose.
Proteinuria: Proteinuria was observed in 2% of patients receiving COMETRIQ, including 1 patient with nephrotic syndrome, vs 0% in placebo. Monitor urine protein regularly during COMETRIQ treatment. Discontinue COMETRIQ in patients who develop nephrotic syndrome.
Reversible Posterior Leukoencephalopathy Syndrome (RPLS): RPLS, a syndrome of subcortical vasogenic edema diagnosed by characteristic finding on MRI, occurred in 1 (<1%) patient. Evaluate for RPLS in patients presenting with seizures, headache, visual disturbances, confusion, or altered mental function. Discontinue COMETRIQ in patients who develop RPLS.
Embryo-Fetal Toxicity: COMETRIQ can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during COMETRIQ treatment and for 4 months after the last dose.
Hypocalcemia: COMETRIQ can cause hypocalcemia. Based on the safety population, hypocalcemia occurred in 52% of patients treated with COMETRIQ, including Grade 3 or 4 in 12% of patients. Monitor blood calcium levels and replace calcium as necessary during treatment. Withhold and resume at a reduced dose upon recovery or discontinue COMETRIQ depending on severity.
ADVERSE REACTIONS
The most commonly reported adverse drug reactions (≥25% and ≥5% difference vs placebo) were diarrhea (63% vs 33%), stomatitis (51% vs 6%), PPE (50% vs 2%), decreased weight (48% vs 10%), decreased appetite (46% vs 16%), nausea (43% vs 21%), fatigue (41% vs 28%), oral pain (36% vs 6%), hair color changes (34% vs 1%), dysgeusia (34% vs 6%), hypertension (33% vs 4%), abdominal pain (27% vs 13%), and constipation (27% vs 6%).
The most common laboratory abnormalities (≥25%) were increased AST (86% vs 35%), increased ALT (86% vs 41%), lymphopenia (53% vs 51%), increased ALP (52% vs 35%), hypocalcemia (52% vs 27%), hypoalbuminemia (43% vs 16%), neutropenia (35% vs 15%), thrombocytopenia (35% vs 4%), hypophosphatemia (28% vs 10%), and hyperbilirubinemia (25% vs 14%).
Increased levels of thyroid-stimulating hormone (TSH) were observed in 57% of patients receiving COMETRIQ (vs 19% receiving placebo).
In clinical trials, the dose was reduced in 79% of patients receiving COMETRIQ compared to 9% of patients receiving placebo. The median number of dosing delays was 1 in patients receiving COMETRIQ compared to 0 in patients receiving placebo. Adverse reactions led to study treatment discontinuation in 16% of patients receiving COMETRIQ and in 8% of patients receiving placebo.
DRUG INTERACTIONS
Strong CYP3A4 Inhibitors: Reduce the dosage of COMETRIQ if concomitant use with strong CYP3A4 inhibitors cannot be avoided. Avoid grapefruit or grapefruit juice.
Strong CYP3A4 Inducers: Increase the dosage of COMETRIQ if concomitant use with strong CYP3A4 inducers cannot be avoided. Avoid St. John’s wort.
USE IN SPECIFIC POPULATIONS
Lactation: Advise lactating women not to breastfeed during treatment with COMETRIQ and for 4 months after the final dose.
Reproductive Potential: Verify the pregnancy status of females of reproductive potential before starting treatment with COMETRIQ. COMETRIQ may impair fertility in females and males of reproductive potential.
Hepatic Impairment: Reduce the COMETRIQ dosage in patients with mild to moderate hepatic impairment. COMETRIQ is not recommended for use in patients with severe hepatic impairment.
INDICATION
COMETRIQ® (cabozantinib) is indicated for the treatment of patients with progressive, metastatic medullary thyroid cancer (MTC).
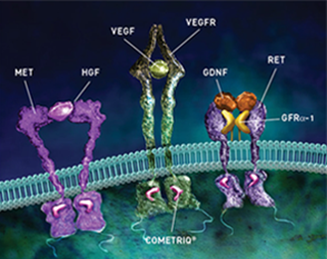
Back to top ^HR=hazard ratio; MET=mesenchymal epithelial transition; OS=overall survival; PFS=progression-free survival; RET=rearranged during transfection; VEGFR=vascular endothelial cell growth factor receptor.
References: 1. COMETRIQ® (cabozantinib) Prescribing Information. Exelixis, Inc,. 2. Elisei R, Schlumberger MJ, Müller SP, et al. Cabozantinib in progressive medullary thyroid cancer. J Clin Oncol. 2013;31(29):3639-3646.